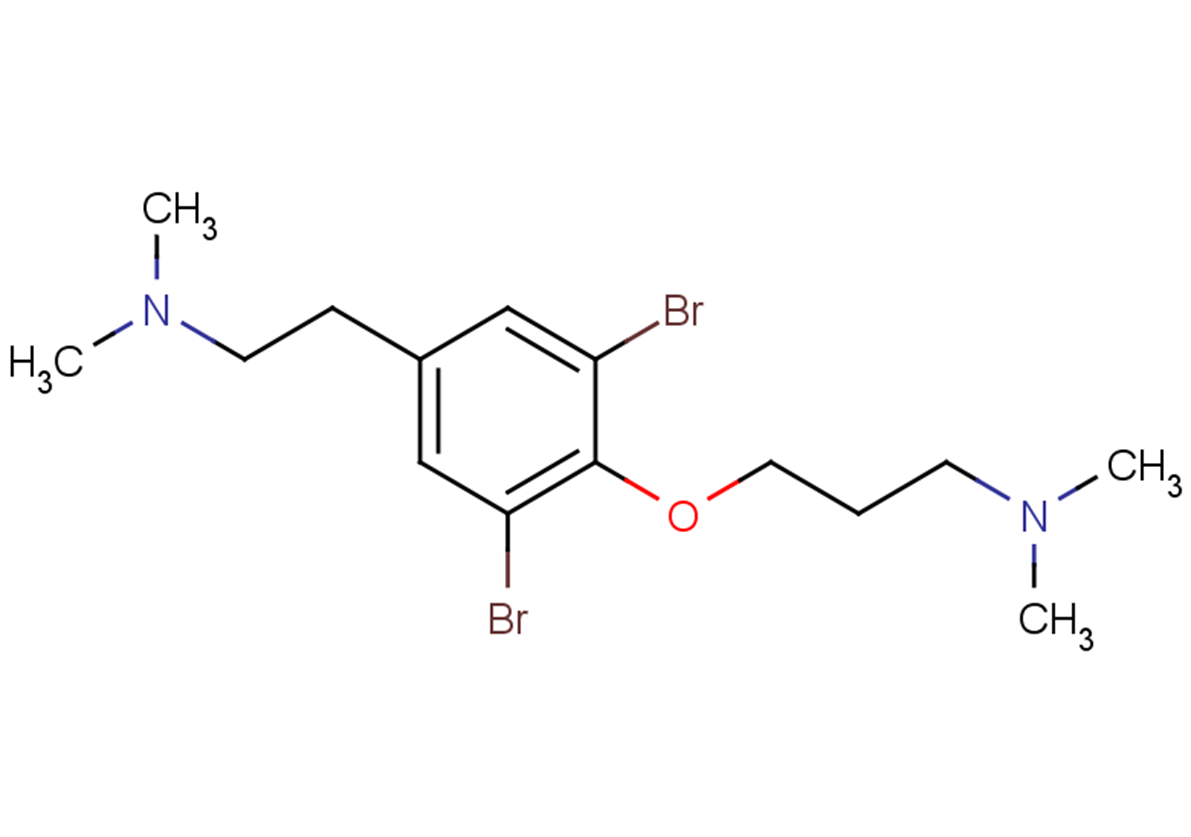
Aplysamine-1
| Code | Size | Price |
|---|
| TAR-T26643-5mg | 5mg | £850.00 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Special offer! Add £1 to your order to get a TargetMol CCK-8 Kit. Read more here. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Quantity:
| TAR-T26643-50mg | 50mg | £1,661.00 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Special offer! Add £1 to your order to get a TargetMol CCK-8 Kit. Read more here. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Quantity:
| TAR-T26643-100mg | 100mg | £2,079.00 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Special offer! Add £1 to your order to get a TargetMol CCK-8 Kit. Read more here. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Quantity:
Prices exclude any Taxes / VAT
Overview
Regulatory Status: RUO
Shipping:
cool pack
Storage:
-20℃
Images
Documents
Further Information
Bioactivity:
Aplysamine-1 is an antagonist of histamine H3 receptor.
CAS:
159026-30-9
Formula:
C15H24Br2N2O
Molecular Weight:
408.178
Purity:
0.98
SMILES:
CN(C)CCCOc1c(Br)cc(CCN(C)C)cc1Br
References
1. Olatunji OJ, Ogundajo AL, Oladosu IA, Changwichit K, Ingkaninan K, Yuenyongsawad S, Plubrukarn A. Non-competitive inhibition of acetylcholinesterase by bromotyrosine alkaloids. Nat Prod Commun. 2014 Nov;9(11):1559-61. PubMed PMID: 25532280.
2. Tian LW, Feng Y, Shimizu Y, Pfeifer T, Wellington C, Hooper JN, Quinn RJ. Aplysinellamides A-C, bromotyrosine-derived metabolites from an Australian Aplysinella sp. marine sponge. J Nat Prod. 2014 May 23;77(5):1210-4. doi: 10.1021/np500119e. Epub 2014 Apr 23. PubMed PMID: 24758268.
3. Tran TD, Pham NB, Fechner G, Hooper JN, Quinn RJ. Bromotyrosine alkaloids from the Australian marine sponge Pseudoceratina verrucosa. J Nat Prod. 2013 Apr 26;76(4):516-23. doi: 10.1021/np300648d. Epub 2013 Mar 14. PubMed PMID: 23489291.
4. Kottakota SK, Evangelopoulos D, Alnimr A, Bhakta S, McHugh TD, Gray M, Groundwater PW, Marrs EC, Perry JD, Spilling CD, Harburn JJ. Synthesis and biological evaluation of purpurealidin E-derived marine sponge metabolites: aplysamine-2, aplyzanzine A, and suberedamines A and B. J Nat Prod. 2012 Jun 22;75(6):1090-101. doi: 10.1021/np300102z. Epub 2012 May 23. PubMed PMID: 22620987.