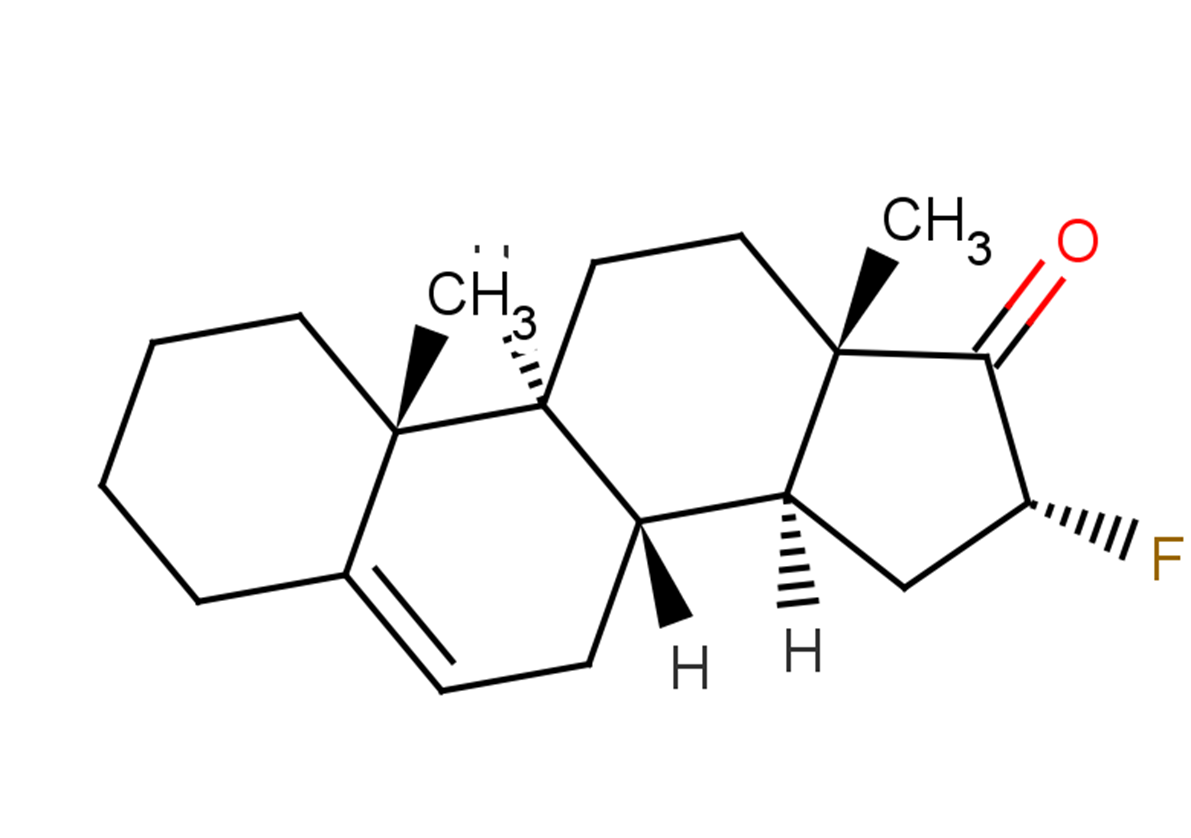
Fluasterone
| Code | Size | Price |
|---|
| TAR-T27338-5mg | 5mg | £850.00 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Special offer! Add £1 to your order to get a TargetMol CCK-8 Kit. Read more here. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Quantity:
| TAR-T27338-50mg | 50mg | £1,661.00 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Special offer! Add £1 to your order to get a TargetMol CCK-8 Kit. Read more here. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Quantity:
| TAR-T27338-100mg | 100mg | £2,079.00 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Special offer! Add £1 to your order to get a TargetMol CCK-8 Kit. Read more here. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Quantity:
Prices exclude any Taxes / VAT
Overview
Regulatory Status: RUO
Shipping:
cool pack
Storage:
-20℃
Images
Documents
Further Information
Bioactivity:
Fluasterone, an NF-κB activation inhibitor, is used potentially for the treatment of metabolic syndrome.
CAS:
112859-71-9
Formula:
C19H27FO
Molecular Weight:
290.422
Purity:
0.98
SMILES:
CC12CCC3C(CC=C4CCCCC34C)C1CC(F)C2=O
References
1. Burgess JP, Green JS, Hill JM, Zhan Q, Lindeblad M, Lyubimov A, Kapetanovic IM, Schwartz A, Thomas BF. Identification of [14C]fluasterone metabolites in urine and feces collected from dogs after subcutaneous and oral administration of [14C]fluasterone. Drug Metab Dispos. 2009 May;37(5):1089-97. doi: 10.1124/dmd.108.023614. PubMed PMID: 19196848; PubMed Central PMCID: PMC2683393.
2. Lindeblad MO, Bauer KS, Zakharov AD, Hill JM, Green JS, Thomas BF, Schwartz A, Kapetanovic IM, Lyubimov A. Pharmacokinetic and tissue distribution study of [14C]fluasterone in male Beagle dogs following intravenous, oral and subcutaneous dosing routes. Chem Biol Interact. 2010 Jan 27;183(2):317-26. doi: 10.1016/j.cbi.2009.10.004. PubMed PMID: 19836365.
3. McCormick DL, Johnson WD, Kozub NM, Rao KV, Lubet RA, Steele VE, Bosland MC. Chemoprevention of rat prostate carcinogenesis by dietary
16alpha-fluoro-5-androsten-17-one (fluasterone), a minimally androgenic analog of dehydroepiandrosterone. Carcinogenesis. 2007 Feb;28(2):398-403. PubMed PMID: 16952912. 4. He Y, Li P, Yalkowsky SH. Solubilization of Fluasterone in cosolvent/cyclodextrin combinations. Int J Pharm. 2003 Oct 2;264(1-2):25-34. PubMed PMID: 12972333.
16alpha-fluoro-5-androsten-17-one (fluasterone), a minimally androgenic analog of dehydroepiandrosterone. Carcinogenesis. 2007 Feb;28(2):398-403. PubMed PMID: 16952912. 4. He Y, Li P, Yalkowsky SH. Solubilization of Fluasterone in cosolvent/cyclodextrin combinations. Int J Pharm. 2003 Oct 2;264(1-2):25-34. PubMed PMID: 12972333.