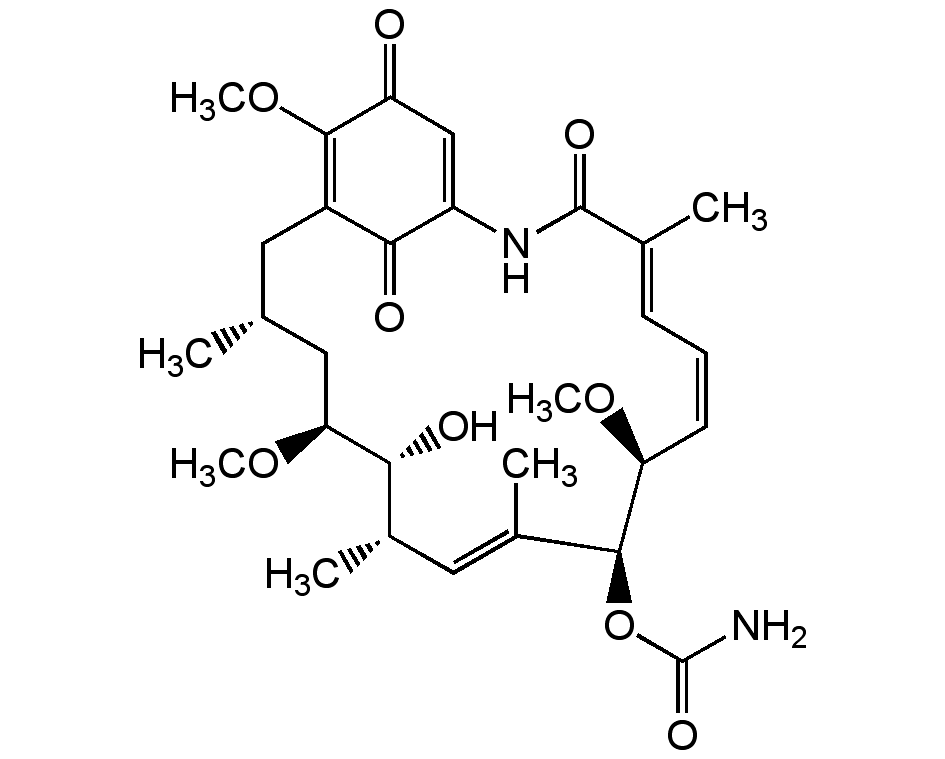
Geldanamycin
| Code | Size | Price |
|---|
| BVT-0196-C100 | 100 ug | £30.00 |
Quantity:
| BVT-0196-C500 | 500 ug | £45.00 |
Quantity:
| BVT-0196-M001 | 1 mg | £70.00 |
Quantity:
| BVT-0196-M005 | 5 mg | £140.00 |
Quantity:
Prices exclude any Taxes / VAT
Overview
Regulatory Status: RUO
Shipping:
20°C
Storage:
-20°C
Images
Documents
Further Information
Alternate Names/Synonyms:
NSC122750; BRN1633093; U-29135
Appearance:
Yellow solid.
CAS:
30562-34-6
EClass:
32160000
Form (Short):
liquid
GHS Symbol:
GHS07
Handling Advice:
Protect from light when in solution.
Hazards:
H302, H312, H319, H332
InChi:
InChI=1S/C29H40N2O9/c1-15-11-19-25(34)20(14-21(32)27(19)39-7)31-28(35)16(2)9-8-10-22(37-5)26(40-29(30)36)18(4)13-17(3)24(33)23(12-15)38-6/h8-10,13-15,17,22-24,26,33H,11-12H2,1-7H3,(H2,30,36)(H,31,35)/b10-8-,16-9+,18-13+/t15-,17+,22?,23?,24-,26+/m1/s1
InChiKey:
QTQAWLPCGQOSGP-GAEFGYQESA-N
Long Description:
Chemical. CAS: 30562-34-6. Formula: C29H40N2O9. MW: 560.6. Isolated from Streptomyces hygroscopicus. Antibiotic. Potent antitumor compound. pp60src tyrosine kinase inhibitor. Inhibits c-mycgene expression in murine lymphoblastoma cells. Inhibits the transforming activity of abl, erbB, fps, src and yes. Binds specifically to heat shock protein 90 (HSP90) and to its endoplasmic reticulum homolog GP96 (GRP94). Destabilizes several oncogene and proto-oncogene products. Potent nuclear hormone receptor family inhibitor.
MDL:
MFCD00274570
Molecular Formula:
C29H40N2O9
Molecular Weight:
560.6
Package Type:
Plastic Vial
Precautions:
P261, P270, P280, P301, P312, P302, P352, P312
Product Description:
Antibiotic. Potent antitumor compound. pp60src tyrosine kinase inhibitor. Inhibits c-mycgene expression in murine lymphoblastoma cells. Inhibits the transforming activity of abl, erbB, fps, src and yes. Binds specifically to heat shock protein 90 (HSP90) and to its endoplasmic reticulum homolog GP96 (GRP94). Destabilizes several oncogene and proto-oncogene products. Potent nuclear hormone receptor family inhibitor.
Purity:
>98% (HPLC)
Signal word:
Warning
SMILES:
CO[C@H]1C[C@H](C)CC2=C(OC)C(=O)C=C(NC(=O)C(C)=CC=C/[C@H](OC)[C@@H](OC(N)=O)C(C)=C[C@H](C)[C@H]1O)C2=O
Solubility Chemicals:
Soluble in methanol or DMSO; insoluble in water.
Source / Host:
Isolated from Streptomyces hygroscopicus.
Transportation:
Non-hazardous
UNSPSC Category:
Natural Products/Extracts
UNSPSC Number:
12352200
Use & Stability:
Stable for at least 1 year after receipt when stored at -20°C. After reconstitution protect from light at -20°C.
References
Geldanamycin, a new antibiotic: C. DeBoer, et al.; J. Antibiot. 23, 442 (1970) | Inhibition of c-myc gene expression in murine lymphoblastoma cells by geldanamycin and herbimycin, antibiotics of benzoquinoid ansamycin group: H. Yamaki, et al.; J. Antibiot. 42, 604 (1989) | Geldanamycin selectively destabilizes and conformationally alters mutated p53: M.V. Blagosklonny, et al.; Oncogene 11, 933 (1995) | Inhibition of the oncogene product p185erbB-2 in vitro and in vivo by geldanamycin and dihydrogeldanamycin derivatives: R.C. Schnur, et al.; J. Med. Chem. 38, 3806 (1995) | Stable and specific binding of heat shock protein 90 by geldanamycin disrupts glucocorticoid receptor function in intact cells: L. Whitesell & P. Cook; Mol. Endocrinol. 10, 705 (1996) | Geldanamycin as a potential anti-cancer agent: its molecular target and biochemical activity: L. Neckers, et al.; Invest. New Drugs 17, 361 (1999), (Review) | Hsp-90-associated oncoproteins: multiple targets of geldanamycin and its analogs: M.V. Blagosklonny; Leukemia 16, 455 (2002), (Review) | Geldanamycin enhances hepatocyte growth factor stimulation of eNOS phosphorylation in endothelial cells: K. Makondo, et al.; Eur. J. Pharmacol. 582, 110 (2007) | HSP90 antagonist, geldanamycin, inhibits proliferation, induces apoptosis and blocks migration of rhabdomyosarcoma cells in vitro and seeding into bone marrow in vivo: E. Lesko, et al.; Anticancer Drugs 18, 1173 (2007) | Inhibition of heat shock protein 90 impairs epidermal growth factor-mediated signaling in gastric cancer cells and reduces tumor growth and vascularization in vivo: S.A. Lang, et al.; Mol. Cancer Ther. 6, 1123 (2007) | Low dose geldanamycin inhibits hepatocyte growth factor and hypoxia-stimulated invasion of cancer cells: F. Koga, et al.; Cell Cycle 6, 1393 (2007) | Hsp90-inhibitor geldanamycin abrogates G(2) arrest in p53-negative leukemia cell lines through the depletion of Chk1: K. Sugimoto, et al.; Oncogene 27, 3091 (2008)
Related Products
| Product Name | Product Code | Supplier | Elmycin B | BVT-0198 | Bioviotica | Summary Details | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Nargenicin A1 | BVT-0204 | Bioviotica | Summary Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Thaxtomin A | BVT-0206 | Bioviotica | Summary Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 17-AAG | BVT-0244 | Bioviotica | Summary Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||