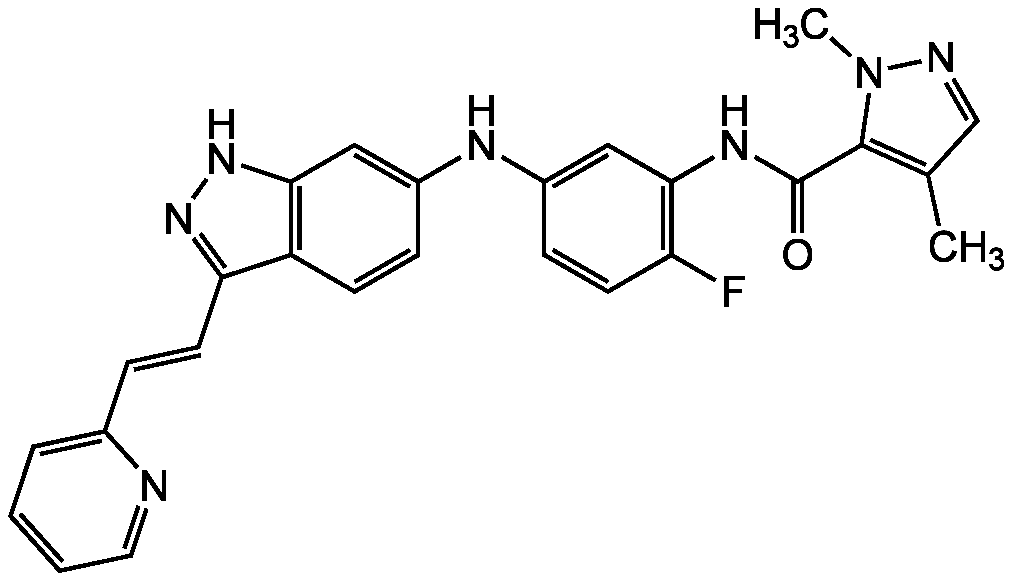
AG-13958
| Code | Size | Price |
|---|
| SYN-1004-M100 | 100 mg | Enquire |
Quantity:
| SYN-1004-M050 | 50 mg | Enquire |
Quantity:
| SYN-1004-M001 | 1 mg | £303.00 |
Quantity:
| SYN-1004-M005 | 5 mg | £590.00 |
Quantity:
| SYN-1004-M010 | 10 mg | £900.00 |
Quantity:
Prices exclude any Taxes / VAT
Overview
Regulatory Status: RUO
Shipping:
-20°C
Images
Documents
Further Information
Alternate Names/Synonyms:
AG-13958
Appearance:
Solid.
CAS:
319460-94-1
EClass:
32160000
Form (Short):
liquid
InChi:
InChI=1S/C26H22FN7O/c1-16-15-29-34(2)25(16)26(35)31-24-14-19(7-10-21(24)27)30-18-6-9-20-22(32-33-23(20)13-18)11-8-17-5-3-4-12-28-17/h3-15,30H,1-2H3,(H,31,35)(H,32,33)/b11-8+
InChiKey:
LLMBCXVXEQNNJB-DHZHZOJOSA-N
Long Description:
Chemical. CAS: 319460-94-1. Formula: C26H22FN7O. MW: 467.5. AG13958 is a VEGF inhibitor that was in clinical development for treatment of age-related macular degeneration (AMD). The drug displays low water solubility and so far in trials delivery has been difficult needing direct injection to be truly effective and it varied by the species used because of species specific differences in eye circulation and vascularization. As a VEGF inhibitor AG13958 was targeted to FLT/TYK receptor inhibition.
Molecular Formula:
C26H22FN7O
Molecular Weight:
467.5
Package Type:
Plastic Vial
Product Description:
AG13958 is a VEGF inhibitor that was in clinical development for treatment of age-related macular degeneration (AMD). The drug displays low water solubility and so far in trials delivery has been difficult needing direct injection to be truly effective and it varied by the species used because of species specific differences in eye circulation and vascularization. As a VEGF inhibitor AG13958 was targeted to FLT/TYK receptor inhibition.
Purity:
>95%
Solubility Chemicals:
Soluble in DMSO.
Transportation:
Non-hazardous
UNSPSC Category:
Protein Kinase Modulators
UNSPSC Number:
12352200
Use & Stability:
Stable for at least 2 years after receipt when stored at -20°C.
References
2nd Ophthalmic Drug Development and Delivery Summit: M. Marra, et al.; Expert Opin. Drug Deliv. 4, 77 (2007)