Alizarin Red S
Product Code:
CDX-A0670
CDX-A0670
Regulatory Status:
RUO
RUO
Shipping:
Ambient
Ambient
Storage:
Short term: +20°C. Long term: +20°C.
Short term: +20°C. Long term: +20°C.
No additional charges, what you see is what you pay! *
| Code | Size | Price |
|---|
| CDX-A0670-G050 | 50 g | £81.00 |
Quantity:
Prices exclude any Taxes / VAT
Stay in control of your spending. These prices have no additional charges to UK mainland customers, not even shipping!
* Rare exceptions are clearly labelled (only 0.14% of items!).
* Rare exceptions are clearly labelled (only 0.14% of items!).
Multibuy discounts available! Contact us to find what you can save.
This product comes from: Switzerland.
Typical lead time: 7-10 working days.
Contact us for more accurate information.
Typical lead time: 7-10 working days.
Contact us for more accurate information.
- Further Information
- Documents
- References
- Show All
Further Information
Appearance:
Brownish yellow powder.
CAS:
130-22-3
EClass:
32160000
Form:
solid
Handling Advice:
Protect from light and moisture.
InChi:
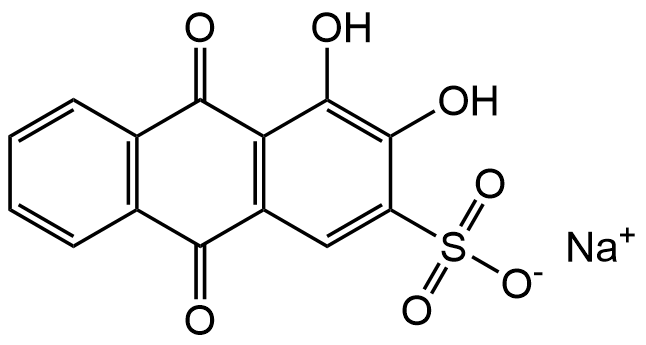
InChI=1S/C14H8O7S.Na/c15-11-6-3-1-2-4-7(6)12(16)10-8(11)5-9(22(19,20)21)13(17)14(10)18;/h1-5,17-18H,(H,19,20,21);/q;+1/p-1
InChiKey:
HFVAFDPGUJEFBQ-UHFFFAOYSA-M
Long Description:
Chemical. CAS: 130-22-3. Formula: C14H7NaO7S. MW: 342.26. Alizarin Red S is a well-established anionic anthraquinone dye widely used as a pH indicator or for the detection and quantification of calcium and other divalent metal ions. It forms a bright red to orange-red complex with calcium salts, making it a standard reagent for histological staining and cell culture assays involving mineralization. In biomedical research, Alizarin Red S is most commonly used to assess osteogenic differentiation by staining calcium deposits in cultured cells and tissue sections. The dye is also employed in analytical chemistry and materials science for metal ion binding studies due to its strong chelation properties. Its strong color response and reliable metal-binding properties make it an excellent choice for osteogenesis studies, histological staining, and spectrophotometric analysis in both research and quality control laboratories. It acts as a colorometric pH indicator. 1st transition range: pH 4.3 - pH 6.3 Lightly yellow - pink/red. 2nd transition range: pH 9.4 - pH 12.0 Brown orange - violet.
MDL:
MFCD00013049
Molecular Formula:
C14H7NaO7S
Molecular Weight:
342.26
Package Type:
Vial
Product Description:
Alizarin Red S is a well-established anionic anthraquinone dye widely used as a pH indicator or for the detection and quantification of calcium and other divalent metal ions. It forms a bright red to orange-red complex with calcium salts, making it a standard reagent for histological staining and cell culture assays involving mineralization. In biomedical research, Alizarin Red S is most commonly used to assess osteogenic differentiation by staining calcium deposits in cultured cells and tissue sections. The dye is also employed in analytical chemistry and materials science for metal ion binding studies due to its strong chelation properties. Its strong color response and reliable metal-binding properties make it an excellent choice for osteogenesis studies, histological staining, and spectrophotometric analysis in both research and quality control laboratories. It acts as a colorometric pH indicator. 1st transition range: pH 4.3 - pH 6.3 Lightly yellow - pink/red. 2nd transition range: pH 9.4 - pH 12.0 Brown orange - violet.
Purity:
>70% (Dye content)
SMILES:
[Na+].Oc1c(O)c(cc2C(=O)c3ccccc3C(=O)c12)S([O-])(=O)=O
Solubility Chemicals:
Soluble in DMSO (10mg/ml) or water (1mg/ml).
Transportation:
Non-hazardous
UNSPSC Category:
Fluorescent Reagents
UNSPSC Number:
41105331
Use & Stability:
Stable for at least 2 years after receipt when stored at RT.
Documents
References
[1] K.F. Adkins; Stain Technol. 40, 69 (1965) | [2] H. Puchtler, et al.; J. Histochem. Cytochem. 17, 110 (1969) | [3] P. Virtanen & K. Isotupa; Acta Anat. 108, 202 (1980) | [4] F. Eggelmeijer, et al.; Clin. Exp. Rheumatol. 9, 11 (1991) | [5] K. Basavaiah, et al.; Talanta 50, 887 (1999) | [6] G. Springsteen & B. Wang; Chem. Commun. 7, 1608 (2001) | [7] I. Sahin & N. Nakiboglu; Anal. Chim. Acta 572, 253 (2006) | [8] S.A. Sangsefedi, et al.; J. Fluoresc. 28, 815 (2018) | [9] W. Ma, et al.; Polymers 11, 165 (2019) | [10] A. Bernar, et al.; Int. J. Mol. Sci. 24, 723 (2022) | [11] C.A.N. Catalan, et al.; Molecules 30, 3286 (2025)