MHC Tetramers
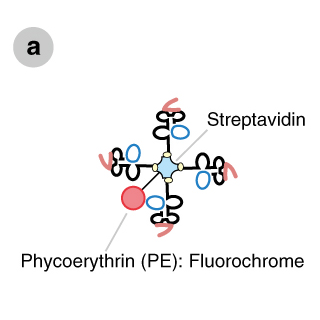
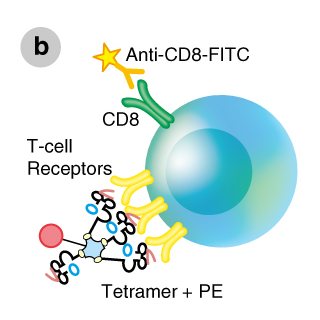
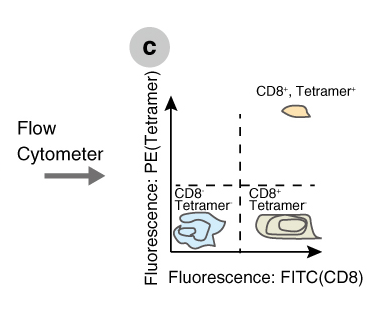
MHC tetramers are complexes of four major histocompatibility complex (MHC) molecules, which are associated with a specific peptide and bound to a fluorochrome-conjugated streptavidin (Figure a). There are two main types of tetramers, Class I and Class II. Class I tetramers bind to a distinct set of T cell receptors (TCRs) on a subset of CD8+ T cells (Figure b), whereas Class II tetramers bind to a distinct population of CD4+ T cells. By mixing tetramers with peripheral blood mononuclear cells (PBMCs) or whole blood and using flow cytometry as a detection system, all CD4+ or CD8+ T cells that are specific for one peptide in the context of a particular MHC allele can be detected (Figures b,c).
MBL provides high-quality MHC Tetramers for the rapid and simple detection of antigen-specific T cells and other types of immune cells by flow cytometry. These tetramers can be used to study several life-threatening diseases, such as infectious diseases, cancer, and autoimmune diseases.
See the Products Available for the Detection of:
Tetramers
Class I MHC Tetramers
CD8+ T cells, also called cytotoxic T lymphocytes (CTLs), recognise complexes of MHC Class I molecules and peptides. CTLs directly kill target cells, including virus-infected and cancer cells.
MBL offers MHC tetramers for various Class I Human, Mouse, Rhesus Macaque and Chicken alleles.
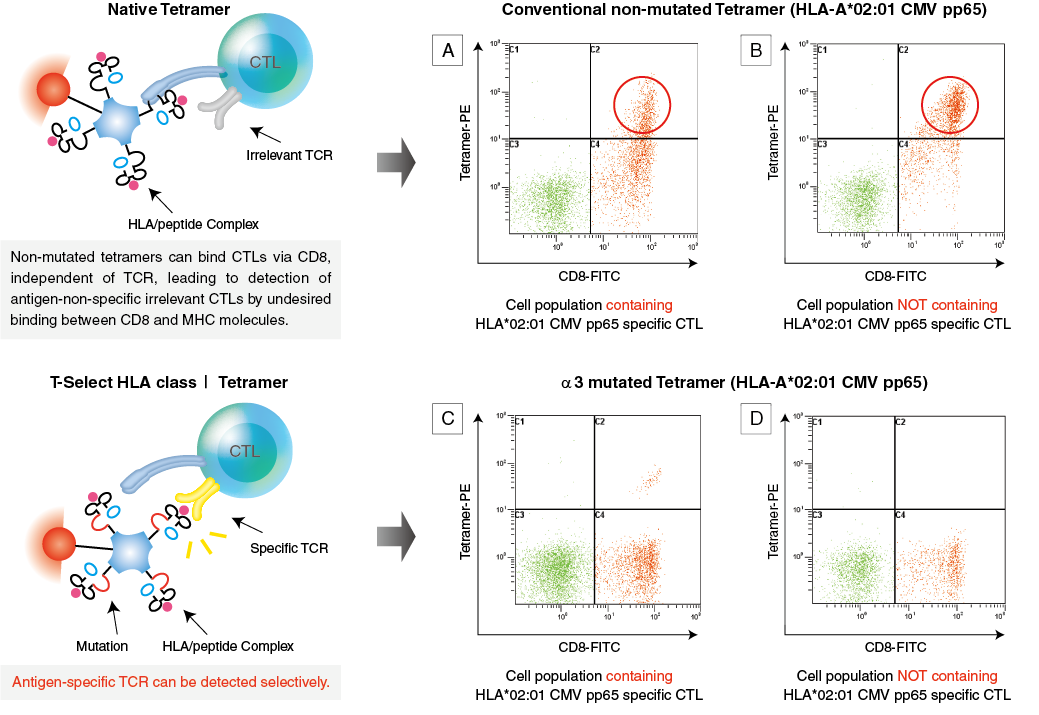
MBL tetramers offer Superior Specificity with α3 Mutation*
The human leukocyte antigen (HLA) system is the name of the MHC in humans. CD8 molecules are known to assist binding of HLA to CTLs in vivo, and thus, HLA molecules have binding sites for CD8 molecules.
Bodinier et al. reported that introducing a mutation (A245V) in the HLA class I heavy chain α3 domain minimised unwanted binding to CD8 molecules and dramatically improved specificity (Nat. Med. 2000, 6: 707).
MBL has incorporated the patented mutation of the α3 domain in their HLA class I tetramers and is the only manufacturer of MHC multimers with this technology to enhance specificity for the antigen-specific T cell receptor.
*Only human and macaque HLA Class I Tetramers contain the alpha 3 mutation.
Class II MHC Tetramers
CD4+ T cells, which include helper and regulatory T cells, recognise complexes of MHC class II molecules and peptides. CD4+ T cells play an important role in health and disease. MHC Class II tetramers are an important tool for investigating rare antigen-specific CD4+ T cells, such as CD4+CD25+ regulatory T cells. In addition to monitoring disease progression and therapeutic intervention, Class II tetramers can be used to study tolerance induction, vaccination efficacy, and autoimmunity.
MBL offers MHC tetramers for Class II Human and Mouse alleles.
See all Class II MHC Tetramers
Non-Classical Tetramers
CD1d
CD1d is a membrane protein non-covalently bonded to β2-microglobulin (β2m), which shows high homology between human and mice. CD1d can present α-galactosylceramide (α-GalCer), a glycolipid extracted and isolated from the marine sponge, and this complex can activate human and murine CD1d-restricted NKT cells.
MBL's CD1d Tetramer-PE is a reagent prepared by tetramerisation of complexes of CD1d and β2m by PE- or APC-labelled streptavidin. Binding this reagent to α-GalCer enables highly sensitive detection of CD1d-restricted NKT cells and can be combined with antibodies to study NKT cell function by flow cytometry.
HLA-E
Like classical MHC molecules, the non-classical MHC Class I molecule HLA-E is involved in self/non-self-recognition and the immune response. However, unlike classical MHCs, it exhibits little polymorphism.
HLA-E inhibits the cellular immune response by binding to the CD94/NKG2 receptor expressed on NK and CD8+ T cells. It is also known to promote activation by binding to the T cell receptor (TCR) expressed on CD8+ T cells.
HLA-E is a key research target for cancer, specifically melanoma and osteosarcoma, and infectious diseases.
MBL offers MHC tetramers against HLA-E targets.
Read more about HLA-E Tetramers
TCR Tetramers
A TCR Tetramer is a reagent prepared by tetramerising biotinylated antigen-specific (TCRs) with the help of phycobiliprotein-labelled streptavidin.
MBL has also efficiently manufactured a TCR Tetramer reagent, which is recognised by MHC, and has successfully commercialised it. Moreover, their cancer antigen-specific (survivin-2B, PBF) TCR Tetramer reagents* have also been assessed for their staining properties by flow cytometry.
*These products are licensed under JP patent No. P2017-169566A.
QuickSwitch™ Kits – Create your own custom MHC Tetramers
Create new specificity tetramers in your lab in just a few hours with QuickSwitch™, a proprietary technology for exchanging peptides on an MHC tetramer.
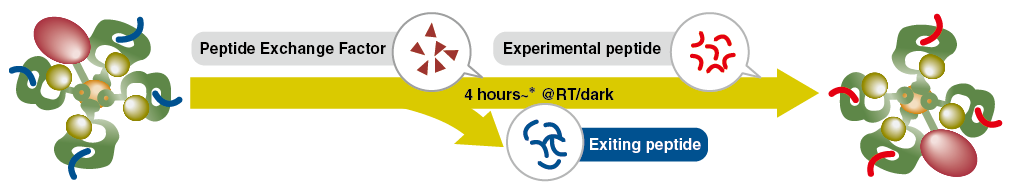
Principle of the Peptide Exchange Reaction
MHC tetramers in QuickSwitch™ Custom Tetramer Kits are pre-bound with “exiting peptide” to maintain structural integrity. Replacing the exiting peptide is achieved by adding a peptide exchange factor and your peptide of interest, resulting in the creation of your custom tetramer (reaction time is ~4 hours*).
*Reaction time is different for each kit.
Determination of Peptide Exchange Efficiency
QuickSwitch™ Quant Tetramer Kits contain an FITC-labelled antibody which detects the exiting peptide pre-bound to MHC molecules. After the peptide-exchange reaction, the MHC tetramers are adsorbed into special beads and react with the antibody for the determination of peptide exchange efficiency by flow cytometry.
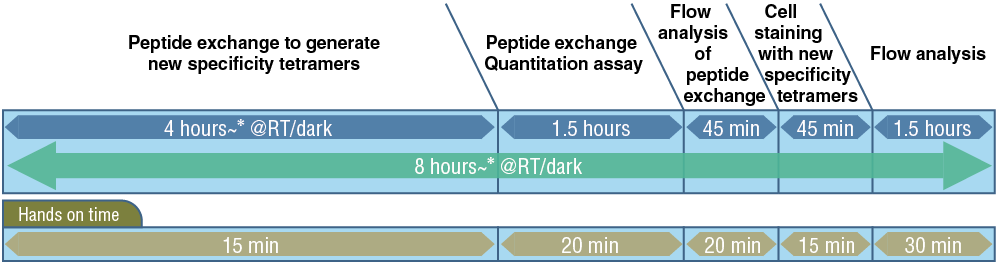
Workflow
Peptide exchange, quantification, cell staining, and flow cytometry analysis can all be performed in one day!
See all QuickSwitchTM Custom Tetramer Kits
QuickSwitch™ Peptide Screening Kits
QuickSwitch™ Peptide Screening Kits are a fresh, new technology that allows for analysis of many peptides in an easy and ready-to-use high-throughput format. There is no need for optimising prior to use. One 96-well plate allows for accurate peptide-MHC binding affinity results that can be gathered in one day.
- High throughput MHC monomer/peptide screening in a 96-well plate assay
- Evaluate immune potentiators or vaccine efficacy
- Neo-epitope screening for further downstream T cell recognition and activation assays
- Determine peptide affinity for specific MHC Class I alleles
- Target discovery of new peptides for immunotherapy and drug discovery
Custom MHC Monomers and Tetramers
MBL are able to manufacture custom MHC tetramers and biotinylated monomers for a range of alleles to your specification.
MBL will first determine feasibility before any production starts. Once feasibility is determined, you can provide your own peptide, or MBL can provide the peptide for you. We will keep you updated with each step of your custom tetramer building process, all while maintaining confidentiality upon request.
Contact us to request a custom MHC Monomer/Tetramer order form and a member of our team will be in touch with you shortly about your request.

